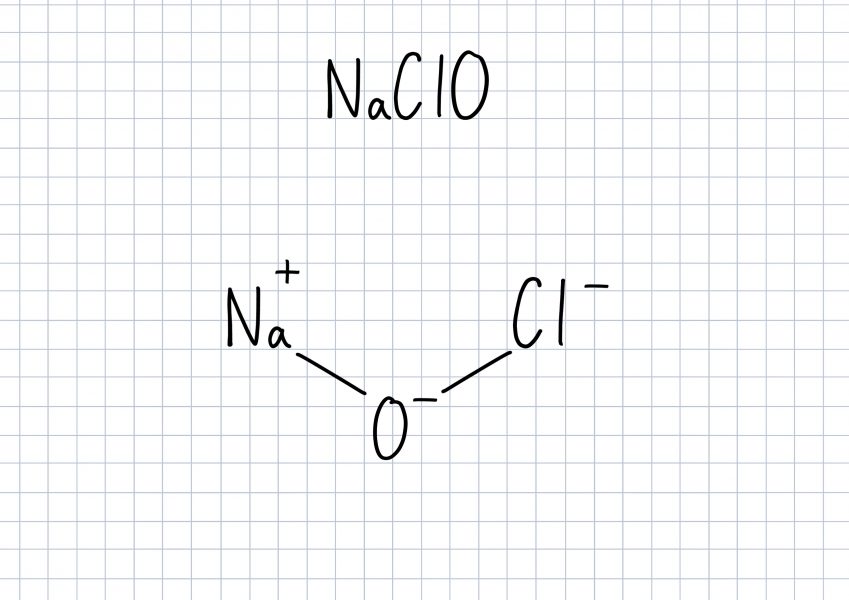
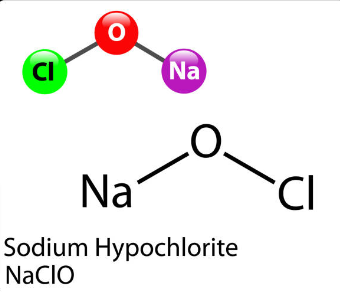
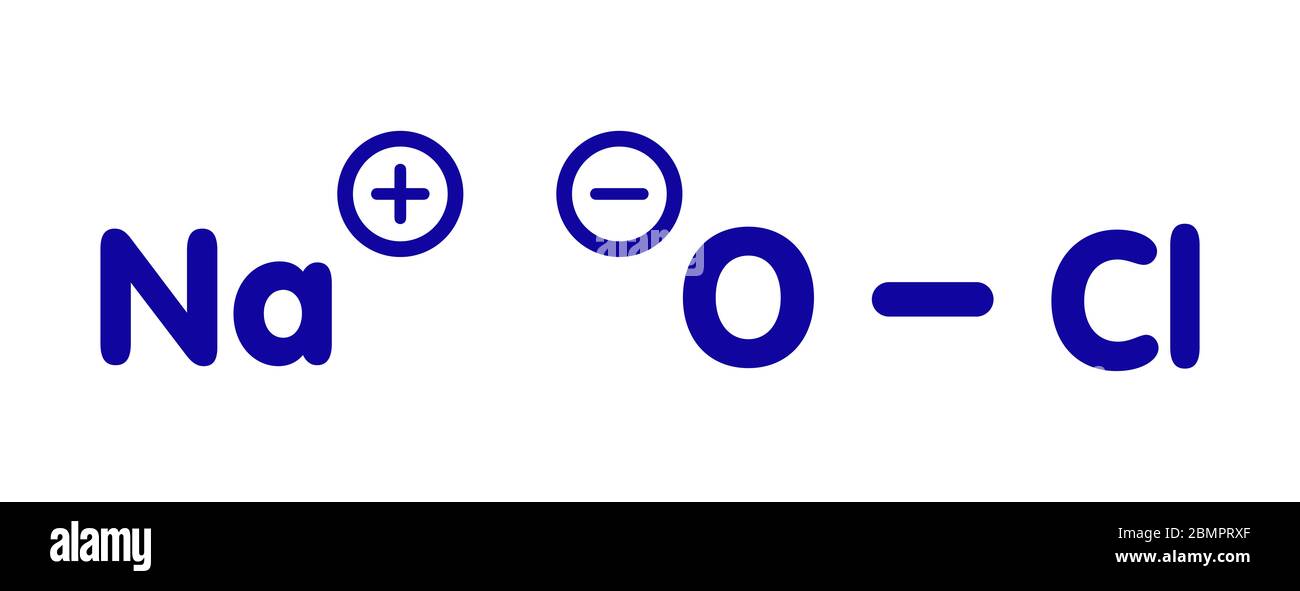NaOCl+Fe+H2O=NaCl+Fe(OH)3 balance the chemical equation @mydocumentary838. naocl+fe+h2o=nacl+fe(oh)3 - YouTube
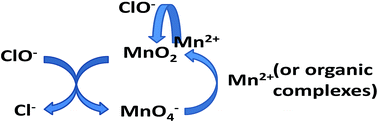
Enhanced removal of manganese in organic-rich surface water by combined sodium hypochlorite and potassium permanganate during drinking water treatment - RSC Advances (RSC Publishing)
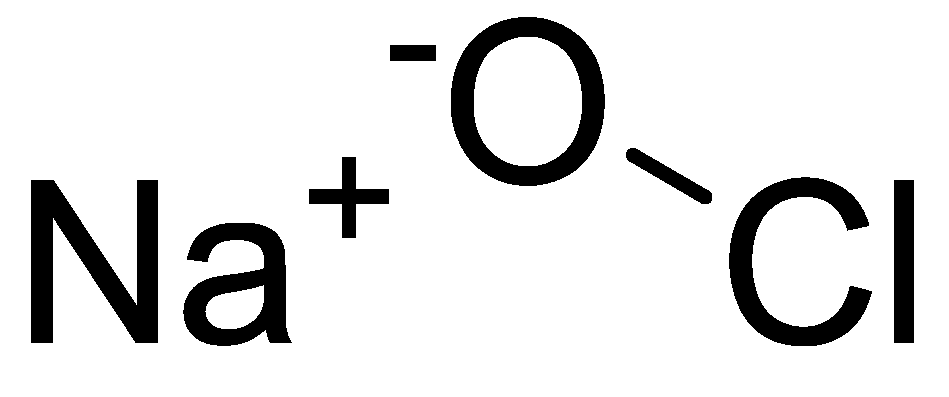
Antibiotics | Free Full-Text | The Interaction of Two Widely Used Endodontic Irrigants, Chlorhexidine and Sodium Hypochlorite, and Its Impact on the Disinfection Protocol during Root Canal Treatment