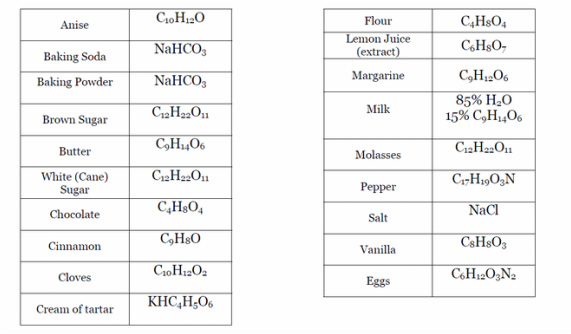
Baking Soda/Vinegar Stoichiometry Lab. Materials Balance Weighing Paper 1 teaspoon of Baking Soda 1 small bottle of vinegar 1 zip lock plastic bag Very. - ppt download

SOLVED: Determine the mass of baking soda (84 g/mol) needed to completely react with 355 mL of vinegar. Assume vinegar is 5% (m/m) acetic acid (60 g/mol) and the density of vinegar

Write the formulae the following and calculate the molecular mass of each one of them.(a) Caustic potash(b) Baking powder(c) Limestone(d) Caustic soda(e) Ethanol(f) Common salt

Write the formulae for the following and calculate the molecular mass for each one of them - YouTube

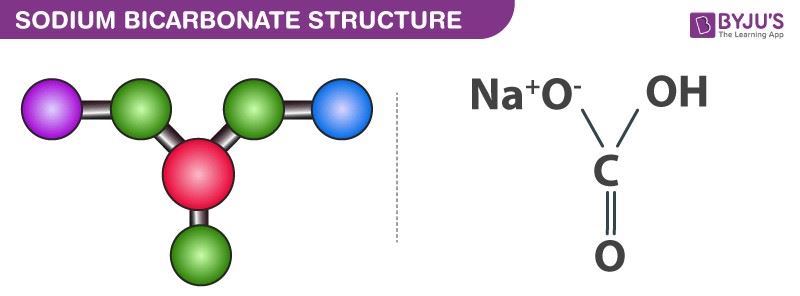





:max_bytes(150000):strip_icc()/sodiumbicarbonate2-599f0a4cb501e800113dd78f.png)